Infectious Disease
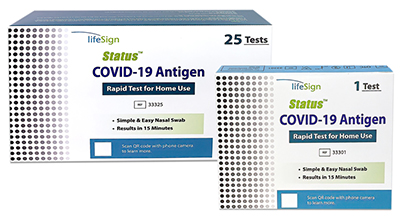
Status™ Covid-19 Antigen Rapid Test For Home Use
Rapid Immunoassay for the Direct Detection of SARS-Cov-2 Antigen for use with anterior nasal specimens for at home use.
FDA Emergency Use Authorization (EUA)
- Visually Read in 15 minutes
- For Home Use, Workplace Testing, Schools
- Ages 14 years and older
- Ages 2 years and older when specimen collected and tested by an adult.
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| Status™ Covid-19 Antigen Rapid Test For Home Use | 33301 | 1 | EUA | ||
| Status™ COVID-19 Antigen Rapid Test For Home Use | 33302 | 2 | EUA | ||
| Status™ COVID-19 Antigen Rapid Test For Home Use | 33325 | 25 | EUA |
Status™ Covid-19/Flu A&B
A Rapid Immunoassay for the Simultaneous Direct Detection and Differential Diagnosis of SARS-CoV-2, Influenza Type A and Type B Antigen from anterior nasal and nasopharyngeal swab specimens
COVID-19
- Anterior nasal swab specimen − Sensitivity 93.8 %, Specificity 100%
- Nasopharyngeal − Sensitivity 93.1 %, Specificity 100%
Flu A - Sensitivity 91.4%, Specificity 95.7%
Flu B - Sensitivity 87.6%, Specificity 95.9%
FDA 510(k) cleared & CLIA Waived
Visually read in 15 minutes
Flocked nasopharyngeal swab for superior specimen collection and patient comfort
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| Status™ Covid-19/Flu A&B | 33225 | 25 | EUA |
BioSign® Flu A&B
An in vitro rapid qualitative test that detects influenza type A and type B antigens directly from nasal swab, nasopharyngeal swab and nasal aspirate/wash specimens
- CLIA WAIVED for nasal swab and nasopharyngeal swab specimens
- Rapid diagnosis for early treatment
- Innovative flip test design
- Easy-to-read cassette
- Pre-measured extraction reagent capsule for increased accuracy and ease of use
- Flocked nasal swabs included for better specimen collection
- Detects H1N1 (Swine Flu)
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® Flu A&B | BSP-510 | 25 | √ | √ | √ |
Status® Strep A – Flip
Rapid Detection of Group A Streptococcal Antigen
Directly From Throat Swab Specimens
- External positive and negative controls included
- Single use reagent capsules
- Positive results in 5 min
- Sensitivity 96%, specificity 99%
- No counting reagent drops – fewer steps for greater accuracy
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| Status® Strep A – Flip | BSP-182 | 25 | √ | √ | √ |
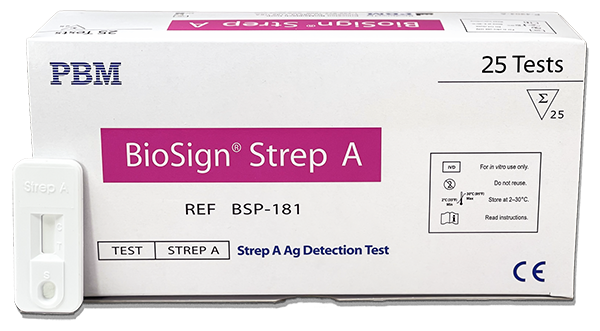
BioSign® Strep A
Rapid Detection of Group A Streptococcal Antigen
Directly From Throat Swab Specimens
- Cassette format
- Simple procedure
- Sensitivity 96.2% , Sensitivity 98.7%
- Results in 5-10 minutes
- External Controls included in kit
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® Strep A | BSP-181 | 25 | √ | √ | √ |
BioSign® H. pylori IgM/IgG/IgA
For the qualitative detection of anti-Helicobacter pylori antibody in human whole blood, serum, or plasma specimens.
- Requires only 25 μl of whole blood or 10 μl serum/plasma
- Single-reagent procedure
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® H. pylori IgM/IgG/IgA | BSP-406WB | 35 | whole blood | √ | √ |
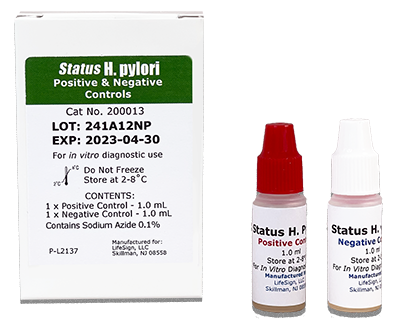
BioSign® H. pylori Control Set
Status H. pylori Control set are external controls to monitor the performance of the Status H.pylori test devices. The set contains a positive and a negative serum controls for qualitative use.
- Liquid- Ready to use
- Convenient plastic dropper tip vials
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® H. pylori Control Set | 200013 | 2 x 1 mL |
BioSign® Rubella IgG
Immunochromatographic assay for the qualitative detection of anti-Rubella Antibody (IgG) in human serum or plasma. Use as an aid in the determination of immune status or for the confirmation of recent rubella infection.
- Rapid results in 15 minutes
- Easy to use – in office testing
- Accurate results
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® Rubella IgG | BSP-171 | 35 | √ | √ |
BioSign® Mono IgM
For the Qualitative Detection of Infectious Mononucleosis Heterophile Antibodies in Whole Blood, Serum or Plasma
- Results within 8 minutes
- FDA cleared with no age restrictions
- Single reagent procedure provides results in 3-8 minutes
- Superior 99.9% sensitivity for earlier IM detection, specificity of 98.8%
- External QC negative and positive controls included
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioSign® Mono IgM | BSP-402WB | 35 | whole blood | √ | √ |
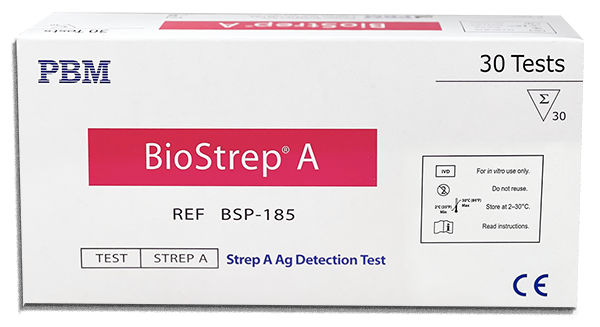
BioStrep® Strep A
Rapid Detection of Group A Streptococcal Antigen Directly from Throat Swab Specimens
- Strip format
- Simple procedure
- Sensitivity 96.2% , Sensitivity 98.7%
- Results in 5-10 minutes
- External Controls included in kit
Ordering Information
| Product | Catalog # | Kit Size | CLIA Waived | FDA | CE |
|---|---|---|---|---|---|
| BioStrep® Strep A | BSP-185 | 25 | √ | √ | √ |
Infectious Disease Products
- Status™ Covid-19 Antigen Rapid Test For Home Use
- Status™ Covid-19/Flu A&B
- BioSign® Flu A&B
- Status® Strep A – Flip
- BioSign® Strep A
- BioSign® H. pylori IgM/IgG/IgA
- BioSign® H. pylori Control Set
- BioSign® Rubella IgG
- BioSign® Mono IgM
- BioStrep® Strep A